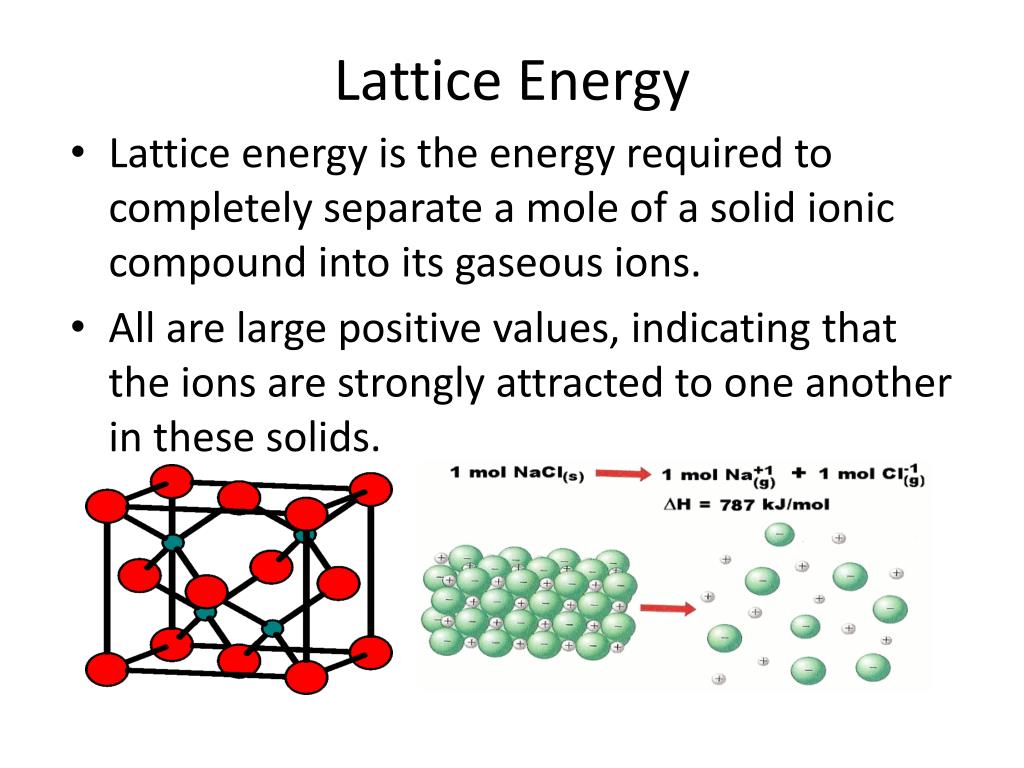
The larger negative value we have for the lattice energy, the more energy released when the lattice was formed, and thus the stronger the lattice structure and the higher the bond order. #DeltaH_"lattice"("SrS") = ?# (cannot find) #"CsI"# ( smallest charge magnitude less similar ionic radii than #"KBr"# larger ionic radii than #"KBr"# as a #pm1# ionic compound).#"KBr"# ( smallest charge magnitude most similar ionic radii smallest ionic radii as a #pm1# ionic compound).#"SrS"# ( highest charge magnitude large difference in ionic radii).So, we expect the highest lattice energy to least lattice energy to be: 2.Between KF K F and LiF L i F (According to Fajans rule KF K F is more ionic than LiF L i F and lattice energy depends directly on ionic character).But, value of lattice energy of KF K F is 808 kJmol1 808 k J m o l 1 and LiF L i F is 1030 kJmol1. The above three methods of comparison are all ordered by their effect on decreasing bond strength. So,according to me MgO M g O should have higher lattice energy but the opposite is true. 
The charge magnitude affects the lattice energy the most by far, followed by the actual ionic radii. #"184 pm"#, #Deltar_("ionic") = "52 pm"#)Īnd finally, in order of largest to smallest charge magnitude, we have: In order of most similar to least similar ionic radii, we have: In order of smallest to largest ionic radii, we have: Now consider these ions on the periodic table: The lattice energy of LiO is the highest among the given compounds as the size of the Li cation is the least among the cations present in the given. Increasing bond order is directly proportional to increasing lattice energy. (Begin with the one with hightest lattice energy) A. 
Question: Place the following in order of decreasing magnitude of lattice energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
